- Home
- Women
- Men
- MSK
- Cardiovascular
- Screening
- About
- Book a Scan
- Blog
Cardiomyopathy is a disease of the heart muscle that affects how well the heart pumps blood. This comprehensive guide explains the five main types of cardiomyopathy, their genetic links, and how an echocardiogram is used to accurately diagnose and monitor the condition.
Secure online booking. Results available within 24 hours.
Cardiomyopathy refers to a group of diseases that affect the heart muscle (myocardium). These conditions cause the heart muscle to become enlarged, thick, or rigid, making it harder for the heart to pump blood effectively to the rest of the body. As the disease progresses, it can lead to heart failure, arrhythmias (irregular heartbeats), and other serious complications.
If you experience sudden, severe breathlessness, chest pain that spreads to your arms or jaw, or if you faint unexpectedly, call 999 or go to your nearest A&E immediately. These can be signs of a life-threatening cardiac event.
Cardiomyopathies are generally classified into two broad categories based on their underlying cause:
Many types of cardiomyopathy have a strong genetic component. For example, familial dilated cardiomyopathy accounts for approximately 29.7% of cases seen in advanced heart failure programmes [4].
If a first-degree relative (parent, sibling, or child) has been diagnosed with cardiomyopathy, clinical guidelines strongly recommend proactive screening with an echocardiogram to detect early signs of the disease before symptoms develop.

Cardiomyopathies are classified based on how the heart muscle's structure and function are altered. The five primary phenotypes each present distinct challenges and require specific diagnostic approaches.

| Type | Structural Changes | Key Characteristics | Prevalence |
|---|---|---|---|
| Hypertrophic (HCM) | The heart muscle (especially the septum) becomes abnormally thick. | Often genetic. Can cause obstruction of blood flow out of the heart (obstructive HCM). A leading cause of sudden cardiac death in young athletes. | 1 in 200 to 1 in 500 people [1]. |
| Dilated (DCM) | The left ventricle becomes enlarged (dilated) and the walls become thin. | The heart muscle weakens and cannot pump effectively (reduced ejection fraction). The most common reason for heart transplantation. | Approximately 1 in 2,500 people [1]. |
| Restrictive (RCM) | The heart muscle becomes rigid and stiff, though not necessarily thickened. | The heart cannot relax and fill with blood properly during diastole. Often caused by infiltrative diseases like cardiac amyloidosis. | Rare; accounts for 2% to 5% of all cardiomyopathies [1]. |
| Arrhythmogenic (ARVC) | Heart muscle tissue in the right ventricle is replaced by fat and scar tissue. | Highly arrhythmogenic, increasing the risk of dangerous irregular heartbeats. Strongly linked to genetic mutations in desmosomal genes. | Rare; primarily affects young adults and athletes. |
| Left Ventricular Non-Compaction (LVNC) | The lower left chamber has a spongy, "trabeculated" appearance. | A rare congenital condition where the heart muscle does not develop normally in utero. Can lead to heart failure and blood clots. | Rare congenital anomaly. |
In the early stages, cardiomyopathy may not cause any noticeable symptoms. However, as the heart's ability to pump blood declines, symptoms of heart failure and arrhythmias typically emerge.
Feeling short of breath, especially during physical exertion or when lying flat in bed. This occurs because fluid backs up into the lungs when the heart cannot pump efficiently.
A profound sense of tiredness that does not improve with rest. This happens because the heart is unable to deliver enough oxygen-rich blood to the body's tissues and muscles.
Noticeable swelling in the legs, ankles, feet, or abdomen. This fluid retention is a classic sign of congestive heart failure associated with dilated or restrictive cardiomyopathy.
Experiencing a fluttering, racing, or pounding heartbeat. In severe cases, arrhythmias can cause dizziness, lightheadedness, or sudden fainting (syncope).
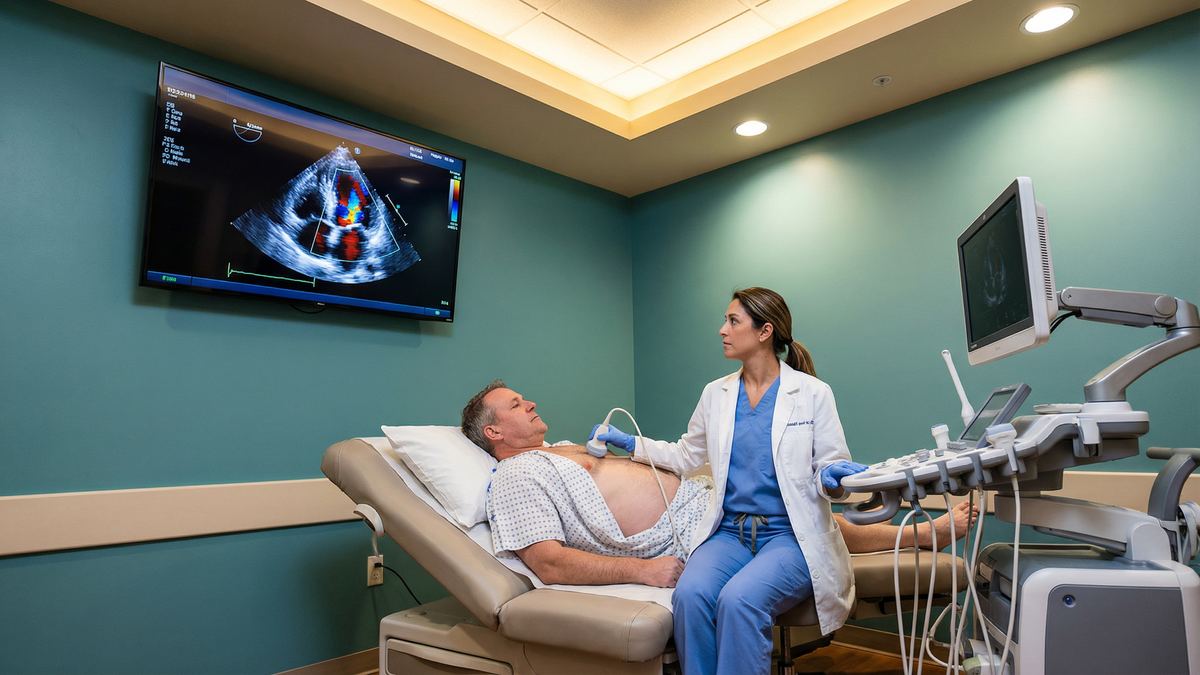
An echocardiogram (ultrasound-scan of the heart) is the primary and most important imaging test used to diagnose and classify cardiomyopathy. It provides real-time, detailed images of the heart's structure and function without any radiation.
During the ultrasound-scan, the sonographer will assess several critical parameters to determine the type and severity of cardiomyopathy:
Cardiac amyloidosis is a primary cause of Restrictive Cardiomyopathy. It occurs when abnormal proteins (amyloid fibrils) deposit in the heart tissue. Real-world data shows that patients often face significant diagnostic delays, with a median of 300 days elapsing from initial suspicion to final confirmation [8].
Advanced echocardiography techniques, such as global longitudinal strain (GLS) imaging, can detect the classic "apical sparing" pattern associated with amyloidosis, facilitating earlier diagnosis and access to targeted therapies like tafamidis [7].